-

Supreme Court Casts Doubt on Trump Birthright Citizenship Order
01:55
-

Young Boy’s Birthday Surprise of WWE Tickets Brings Him to Tears
01:03
-
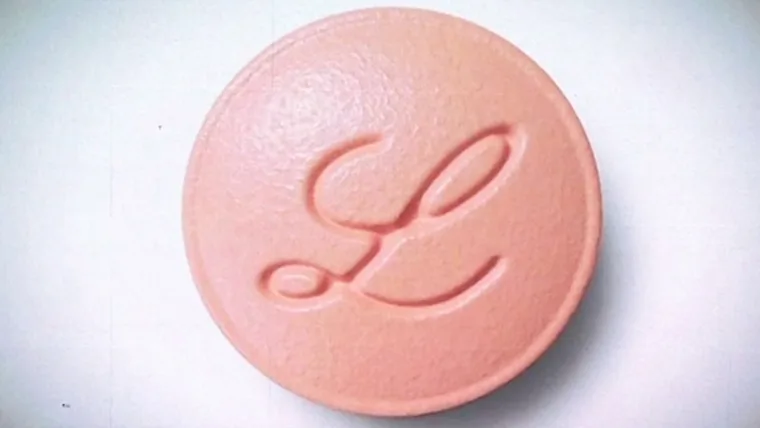
Now Playing

FDA Approves Eli Lilly’s New Weight Loss Pill Foundayo
03:12
-
UP NEXT

Hawaii Doctor Takes the Stand in Attempted Murder Trial
02:19
-

Tiger Woods Granted Permission to Leave US for Treatment
00:27
-

Trump Vows to Hit Iran ‘Extremely Hard’ in Coming Weeks
05:50
-

Artemis II Roars Into Space in Groundbreaking Moon Mission
05:37
-

Hershey’s to Return to Pure Chocolate Ingredients in All Recipes
00:32
-

Influencer Shines Light on Struggling Family-Owned Restaurant
04:56
-

Get an Inside Look at How Fiesta Tableware Products Are Made
05:00
-

More Americans Are Taking on Side Hustles to Make Extra Cash
04:06
-

Why Las Vegas Hotels Are Rolling the Dice on All-Inclusive Options
03:10
-

A Look Back at Apple’s History and Impact as It Marks 50 Years
05:40
-

Trump to Attend Supreme Court Birthright Citizenship Hearing
01:50
-

Son of Hawaii Doctor Accused of Trying to Kill Wife Testifies in Trial
00:45
-

Hegseth Lifts Suspension of Crew Who Flew by Kid Rock’s Home
00:29
-

Judge Halts Construction on Trump White House Ballroom Project
00:41
-

8-Year-Old Gets Warmest Welcome From Her Class After Surgery
00:50
-

Tiger Woods Breaks His Silence Following DUI Arrest in Florida
02:51
-

Trump Says War With Iran Will End Soon Even as Attacks Escalate
02:10
-

Supreme Court Casts Doubt on Trump Birthright Citizenship Order
01:55
-

Young Boy’s Birthday Surprise of WWE Tickets Brings Him to Tears
01:03
-
Now Playing

FDA Approves Eli Lilly’s New Weight Loss Pill Foundayo
03:12
-
UP NEXT

Hawaii Doctor Takes the Stand in Attempted Murder Trial
02:19
-

Tiger Woods Granted Permission to Leave US for Treatment
00:27
-

Trump Vows to Hit Iran ‘Extremely Hard’ in Coming Weeks
05:50
-

Artemis II Roars Into Space in Groundbreaking Moon Mission
05:37
-

Hershey’s to Return to Pure Chocolate Ingredients in All Recipes
00:32
-

Influencer Shines Light on Struggling Family-Owned Restaurant
04:56
-

Get an Inside Look at How Fiesta Tableware Products Are Made
05:00
-

More Americans Are Taking on Side Hustles to Make Extra Cash
04:06
-

Why Las Vegas Hotels Are Rolling the Dice on All-Inclusive Options
03:10
-

A Look Back at Apple’s History and Impact as It Marks 50 Years
05:40
-

Trump to Attend Supreme Court Birthright Citizenship Hearing
01:50
-

Son of Hawaii Doctor Accused of Trying to Kill Wife Testifies in Trial
00:45
-

Hegseth Lifts Suspension of Crew Who Flew by Kid Rock’s Home
00:29
-

Judge Halts Construction on Trump White House Ballroom Project
00:41
-

8-Year-Old Gets Warmest Welcome From Her Class After Surgery
00:50
-

Tiger Woods Breaks His Silence Following DUI Arrest in Florida
02:51
-

Trump Says War With Iran Will End Soon Even as Attacks Escalate
02:10
Source: Read Full Article













